Nuclear Complex Co-IP Kit Overview
Co-immunoprecipitation (Co-IP) is often used to study the relationships between nuclear protein complexes and nuclear processes such as intracellular signaling and transcriptional regulation. However, because these interactions are often labile, and the proteins & their modifications can be fragile, traditional methods to isolate and study DNA-binding proteins often lead to disruption of nuclear complexes and a loss of protein integrity. Active Motif’s Nuclear Complex Co-IP Kit improves Co-IP by providing a gentle extraction procedure and optimized immunoprecipitation reagents that enhance the recovery of intact nuclear protein complexes.
Note that the antibody binding beads are NOT included in the Nuclear Complex Co-IP Kit and must be supplied by the user. Active Motif recommends the Protein G Agarose Columns (Cat No. 53037/53039) for easy and efficient capture of the IP complex. The Protein G Agarose Columns contain pre-washed protein G agarose beads that have been specifically engineered to reduce non-specific binding. The beads are provided in ready to use filtration columns that streamline the antibody binding, wash, and elution steps.
Nuclear Complex Co-IP Kit Highlights
- Convenient: Simple and easy to use protocol
- Gentle: Extraction procedure preserves protein complexes without disrupting protein-protein interactions
- Flexible: Includes Immunoprecipitation reagents of varying stringency to enable the optimization of protein-protein interactions of varying strength
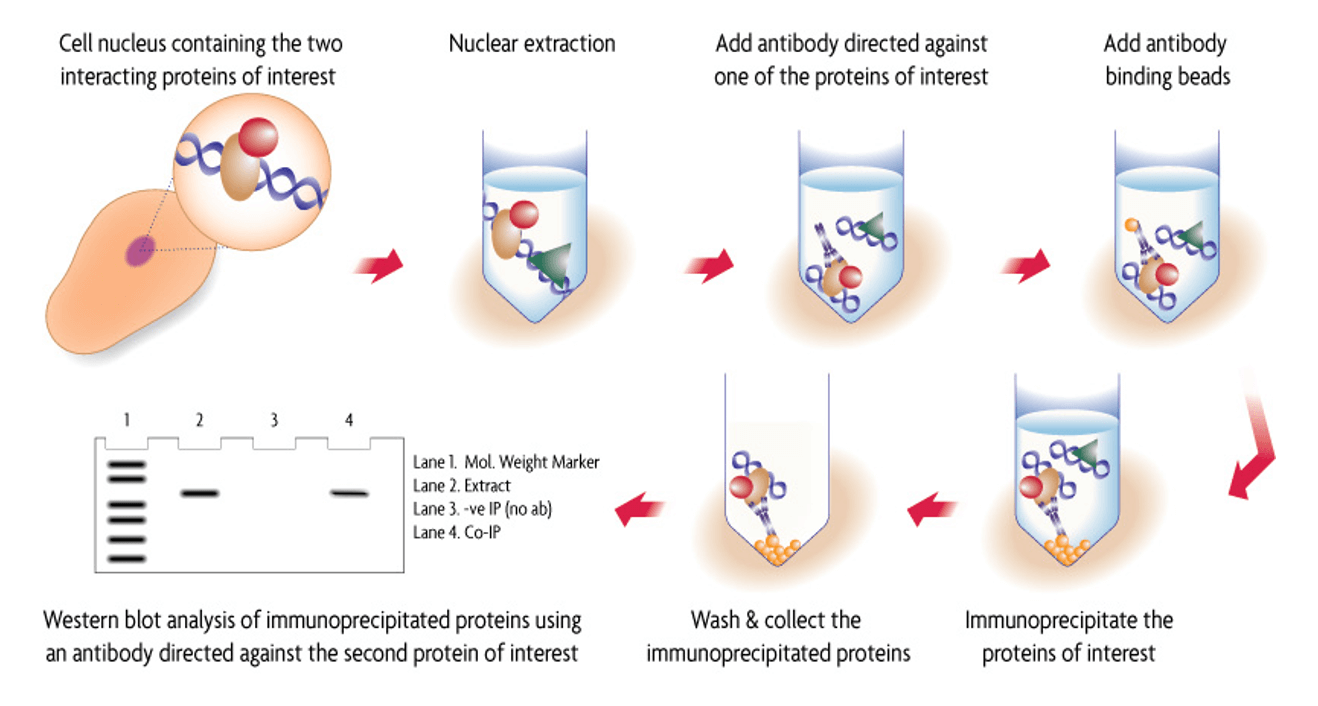
Flow chart of the Co-Immunoprecipitation procedure used in the Nuclear Complex Co-IP Kit.
Nuclear extract is prepared using a combination of low-salt buffers and enzymatic shearing, which helps protect nuclear protein complexes as they are released from the DNA. Immunoprecipitation is then carried out and the protein complex is washed using buffers that can be adjusted by addition of salt and detergent to optimze the stringency required to main the complex while eliminating non-specific proteins. Western blot is then performed using a 2nd antibody directed against a 2nd protein of interest.
Nuclear Complex Co-IP Kit Contents
The Nuclear Complex Co-IP Kit is shipped on dry ice and contains reagents with multiple storage temperatures inside. Please store components according to the storage conditions in the manual after first use. All reagents are guaranteed stable for 6 months from date of receipt when stored properly.
- Protease Inhibitor Cocktail (PIC); Store at -20°C
- 100 mM PMSF; Store at -20°C
- Enzymatic Shearing Cocktail; Store at -20°C
- 1 M DTT; Store at -20°C
- 10X Hypotonic Buffer; Store at 4°C
- Phosphatase Inhibitors; Store at 4°C
- 10X PBS; Store at 4°C
- 0.5 M EDTA; Store at 4°C
- Digestion Buffer; Store at 4°C
- 5X IP High Buffer; Store at 4°C
- 5X IP Low Buffer; Store at 4°C
- BSA; Store at 4°C
- Detergent; Store at RT
- 5 M NaCl; Store at RT
Nuclear Complex Co-IP Kit FAQs
What is the difference between the Nuclear Complex Co-IP Kit (Cat No. 54001) and the Universal Magnetic Co-IP Kit (Cat No. 54002)?
Below are the main differences:
| Nuclear Complex Co-IP Kit (Cat No. 54001) | Universal Magnetic Co-IP kit (Cat No. 54002) |
|---|---|
| Processes nuclear lysates | Processes nuclear and whole-cell lysates |
| More customizable offering varying stringencies | Easier to use |
| Uses Protein G agarose columns |
Uses magnetic beads (included in kit) |
If I am switching from low to high stringency buffer, do I have to switch for both the IP buffer and the wash buffer, or can I switch for one but not the other?
The Nuclear Complex Co-IP Kit is very customizable kit in terms of stringency. Both Low and High IP buffers are compatible with each other, so you are welcome to use either the same buffer for preparing the IP incubation buffer (Day 1) and IP wash buffer (Day 2) or mix and match them to your specific needs.
Is the difference between the low and high IP stringency buffers, just the detergents and the salt concentration?
The 5X IP High IP and 5X IP Low buffers use different bases (the Low Buffer is HEPES based, whereas the High Buffer is Tris-based), but they have the same salt and detergent composition.
Is this kit compatible with downstream mass spectrometry (MS/LC-MS)?
Active Motif has not used this kit with downstream mass spectrometry. However, there are many publications where it has been used for this application. Various MS instruments are sensitive to different buffer components. Please consult with your instrument’s manual or core facility to determine any components to avoid and we can let you know if they are present in our buffer systems.
Nuclear Complex Co-IP Kit Publications
Search our database of customer publications that have used our Nuclear Complex Co-IP Kit.
Nuclear Complex Co-IP Kit Documents
You might also be interested in:
| Name | Format | Cat No. | Price | |
|---|---|---|---|---|
| Nuclear Complex Co-IP Kit | 50 rxns | 54001 | $425 | Buy |
| Protein G Agarose Columns | 30 rxns | 53039 | $415 | Buy |
| 5 rxns | 53037 | $100 | Buy | |
| 5X IP High Buffer | 25 ml | 37510 | $130 | Buy |
| 5X IP Low Buffer | 25 ml | 37511 | $130 | Buy |
| Detergent | 25 ml | 37517 | $130 | Buy |
Co-Immunoprecipitation (Co-IP) is a powerful method used to study protein/protein interactions. In Co-IP, one antibody is used to immunoprecipitate a target antigen and also co-precipitate any bound interacting proteins within a sample. This complex is then detected by Western blot using a second antibody targeted against one of the bound interacting proteins. However, traditional Co-IP methods are not optimal for studying DNA-binding protein complexes as these complexes are often disrupted during the extraction process. In addition, unstable protein complexes are frequently affected by the salt and detergent composition of the buffers used in the immunoprecipitation process. The Nuclear Complex Co-IP Kit extraction and immunoprecipitation reagents were optimized to help maintain nuclear protein complexes, providing you with the best results possible.
The Nuclear Complex Co-IP method
In the Nuclear Complex Co-IP Kit method, nuclear extracts are prepared by collecting cells in ice-cold PBS with Phosphatase Inhibitors. Then, the cells are resuspended in Hypotonic Buffer to swell the cell membrane and make it fragile. Addition of Detergent causes leakage of the cytoplasmic proteins into the supernatant. After collection of the cytoplasmic fraction, the nuclei are lysed and the nuclear proteins are recovered in a low-salt buffer in the presence of the Protease Inhibitor Cocktail and PMSF. This is followed by the addition of a proprietary Enzymatic Shearing Cocktail. The use of low-salt buffers protects protein complexes in the nucleus; DNA digestion allows a gentle release of undissociated protein complexes from the DNA.
After the protein complexes are collected, an immunoprecipitation reaction is carried out to detect the bound proteins. Two different immunoprecipitation buffers with either a low or high stringency starting composition are provided. In addition, detergent and salt are provided separately to enable you to vary the salt and detergent concentrations. The addition of salt and detergent is ideal for use with robust protein/protein interactions because such conditions reduce background. However, as unstable protein complexes may not withstand high stringencies, the kit format enables stringency to be modified as required by each complex.
Figure 1: Flow chart of the Co-Immunoprecipitation procedure used in the Nuclear Complex Co-IP Kit.
Nuclear extract is prepared using a low-salt buffer and enzymatic shearing, which protects nuclear protein complexes and releases them from the DNA. Immunoprecipitation is then carried out and the protein complex is washed using buffers that whose stringency can be adjusted by addition of salt and detergent, which helps maintain the complex integrity while eliminating non-specific proteins. Western blot is then performed using a 2nd antibody directed against a 2nd protein of interest.
| Co-IP Kit: Buffer Preparation and Recommendations | ||||
|---|---|---|---|---|
| Approach | IP Buffer | 5M NaCl | Detergent | 5M NaCl for Wash Buffer |
| 1. No Supplements | 1X High | N/A | N/A | N/A |
| 1X Low | N/A | N/A | N/A | |
| 2. Addition of Salt | 1X High | 150 mM Final | N/A | N/A |
| 1X Low | 150 mM Final | N/A | N/A | |
| 3. Addition of salt and detergent | 1X High | 150 mM Final | 1% Final | N/A |
| 1X Low | 150 mM Final | 1% Final | N/A | |
| 4. Addition of salt and detergent, and extra salt for final Wash Step | 1X High | 150 mM Final | 1% Final | 300 mM Final |
| 1X Low | 150 mM Final | 1% Final | 300 mM Final | |
Contents & Storage
Please note that the Nuclear Complex Co-IP Kit is shipped on dry ice and contains reagents with multiple storage temperatures inside. Please store each component at the temperature indicated below. All reagents are guaranteed stable for 6 months from date of receipt when stored properly. Each Nuclear Complex Co-IP Kit supplies sufficient reagents to perform 50 co-immunoprecipitation experiments. This kit includes the following components:
- Protease Inhibitor Cocktail (PIC); Store at -20°C
- 100 mM PMSF; Store at -20°C
- Enzymatic Shearing Cocktail; Store at -20°C
- 1 M DTT; Store at -20°C
- 10X Hypotonic Buffer; Store at 4°C
- Phosphatase Inhibitors; Store at 4°C
- 10X PBS; Store at 4°C
- 0.5 M EDTA; Store at 4°C
- Digestion Buffer; Store at 4°C
- 5X IP High Buffer; Store at 4°C
- 5X IP Low Buffer; Store at 4°C
- BSA; Store at 4°C
- Detergent; Store at RT
- 5 M NaCl; Store at RT
Some select publications are listed below.
- “ALS Associated Mutations in Matrin 3 Alter Protein-Protein Interactions and Impede mRNA Nuclear Export.” by Boehringer et al. (2016) Scientific Reports 7(1):14529.


