Jump to your topic of interest:
Available Products
To view complete details, including ordering information, please use the links below.
- Chromeo™ 488 Tools for GSDIM Microscopy
- Chromeo™ 505 Tools for GSDIM Microscopy
- Chromeo™ 546 Tools for GSDIM Microscopy
- ATTO 532 (GSD) secondary antibody conjugates
- ATTO 647N (STED/GSD) secondary antibody conjugates
- Rhodamine 6G (GSD) secondary antibody conjugates
How GSDIM microscopy overcomes the Abbe limit
In regular Fluorescent Microscopy, image resolution is limited by diffraction to approximately half the wavelength of the emitted light. This means that fluorescent dyes, which are frequently closer to one other than about 200 nm when used for staining, can not be optically separated. As shown in Figure 1A, none of the dyes within the excited area will be recognized as individual signals; their fluorescent signals simply overlap.
In Ground State Depletion with Individual Molecule return (GSDIM) microscopy, the majority of dye molecules within the excited area are transformed into a dark state so that only single dye molecules that are well separated from one another are able to emit fluorescence (Figure 1B). Although these well-separated fluorophores are still observed as diffraction-limited spots, the center of these spots resembles the original position of the fluorophore within nanometer accuracy. By producing several thousand images that capture individual fluorescing molecules (Figures 1 B + C), then calculating the center position of these signals, the original position of the fluorophores can be determined. Based on the knowledge of the positions of the individual fluorophores, a super-resolution image is mathematically re-constructed with a resolution as low as 20 nm.
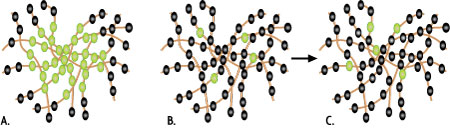
Figure 1: Principle of GSDIM microscopy.Illustration of the exposed area of a sample in classical widefield microscopy (A) or in GSDIM microscopy (B + C) in which only a small number of single dye molecules emit fluorescence. The spheres represent individual dye molecules in fluorescent (green) or “off” mode (black).
To switch the excited dye molecules into a dark (non-fluorescent) state, the GSDIM microscope contains high power laser systems. As indicated in the Jablonski diagram in Figure 2, the laser transforms the molecules to the excited electronic state S1 and to the dark D or triplet state T1. As the lifetime of these “off” states is much higher than the lifetime of the excited state (milliseconds versus nanoseconds), most of the molecules will accumulate in the non-fluorescent dark state. Stochastically, some molecules turn back to the ground state S0 where they will be excited and either allowed to emit fluorescence or switched again into the dark state. As a result of this cycling between the “off” and “on” states, individual molecules are able to emit fluorescent photons. These emitting single dye molecules can be detected as blinking spots in an image sequence.
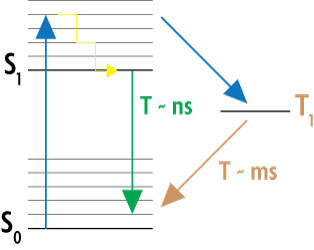
Figure 2: Simplified Jablonski diagram of the GSDIM method.
Principle of the GSD microscope
With GSDIM (Ground State Depletion with Individual Molecule return) microscopy it is possible to exceed the Abbe limit and achieve resolution down to 20 nm. The resolution afforded by GSDIM microscopy facilitates the separation of sub-cellular structures that previously could not be resolved, and increases the confidence in the biological roles of proteins and structures that co-localize. The SR GSD microscope is based on the fully automated Leica AM TIRF MC system that combines high-resolution microscopy with high-end widefield and TIRF (Total Internal Reflection Fluorescence) microscopy. The inverted microscope contains three powerful lasers to stimulate the dyes to their excited or dark triplet states. In addition, the 488, 532 and 642 nm laser systems offer maximum flexibility by enabling the use of a wide range of fluorescent dyes, or combinations of dyes for multi-color approaches. The high resolution image is the result of the overlay of thousands of single frames, whose formation can be monitored “online”. The complete acquisition of than image with a 20-40 nm resolution takes about 2-10 minutes each sample. For more complete information on GSDIM microscopy, please visit the Leica Microsystems website.
Optimized sample preparation for GSDIM microscopy
The sample preparation for GSDIM microscopy is based on standard immunostaining techniques, although it requires some specific optimization. The ability to switch dyes into the dark triplet state is mainly dependent on the use of high energy laser systems for excitation and the use of reducing conditions in the embedding medium. As different dyes interact differently with the surrounding media, the embedding should be changed depending on the dye. Leica recommends Glucose-Oxidase Mix (containing glucose, glucose oxidase and catalase in a PBS buffer of pH = 7.4), PVA-based (Polyvinyl alcohol), MEA-based (β-Mercaptoethylamine) or Mowiol-based embedding media.
For use with its SR GSD microscope, Leica Microsystems has tested and verified the use of Active Motif’s Chromeo™ 488, Chromeo™ 505 and Chromeo™ 546 fluorescent dyes and secondary antibody conjugates. In addition, Leica recommends Active Motif’s Rhodamine 6G (GSD) secondary antibody conjugates, ATTO 532 (GSD) secondary antibody conjugates and ATTO 647N (STED/GSD) secondary antibody conjugates for use with its instrument because they meet the specifications required for GSDIM microscopy. These highly cross-adsorbed secondary antibody conjugates have been optimized for use in high-resolution microscopy, for example by determining the best “dye-to-protein ratio” needed to achieve the maximum image contrast and specific fluorescence intensity.
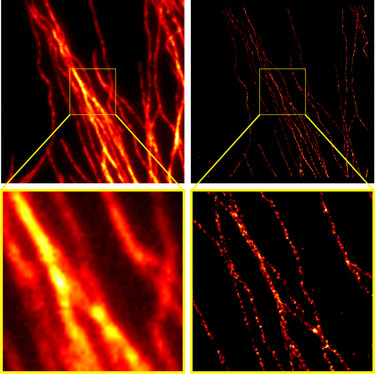
Figure 3: Comparison of conventional widefield microscopy and GSDIM microscopy.Tubulin was stained with a primary monoclonal mouse antibody and with Chromeo 488 Goat anti-mouse IgG (Catalog No. 15031) secondary antibody (left). The widefield (left) and GSDIM (right) images are courtesy of Leica Microsystems, Germany. The "zoomed in" images at the bottom illustrate the enhanced resolution provided by GSDIM.

