Co-Immunoprecipitation (Co-IP) is a powerful method used to study physiologically relevant protein-protein interactions within cell or tissue lysates. In standard Immunoprecipitation (IP) an antibody is used to target and bind with a specific protein in a sample. This protein-antibody immune complex is then captured by antibody binding beads, followed by washing and target protein elution.
Co-IP is similar to IP in that an antibody is used to immunoprecipitate a target protein. However, the goal is to also to co-precipitate any other interacting proteins with it. Once the protein complexes have been isolated, SDS-PAGE and Western blot can be used to detect any one of the bound, interacting proteins using a second targeting antibody.
Traditional methods for performing Co-IP are not optimal for studying DNA-binding protein complexes as they are often disrupted during the extraction process. In addition, many unstable protein complexes can be affected by the salt and detergent composition of the buffers used during immunoprecipitation. Active Motif’s Co-IP Kits have been optimized, using a gentle nuclear or cytoplasmic extraction followed by low-stringency Co-IP and wash buffers to help maintain intact protein complexes.
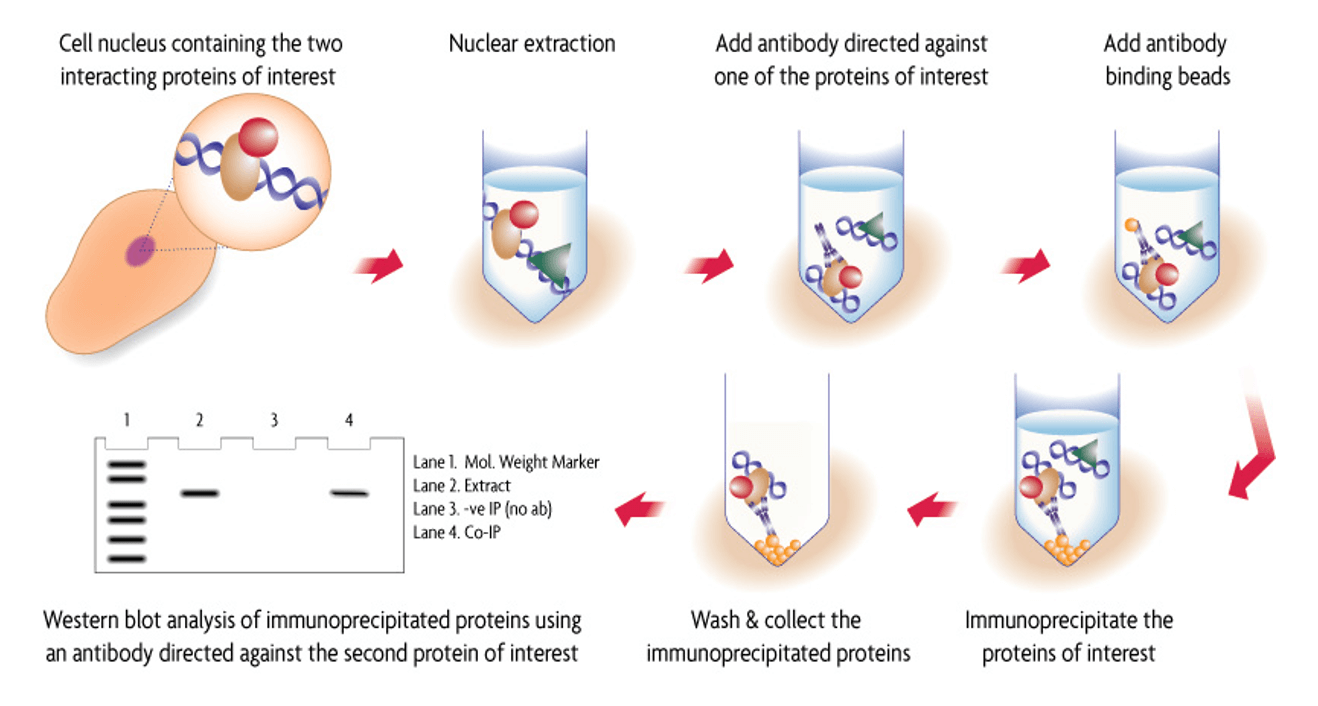
This kit provides reagents for a gentle nuclear extraction and optimized immunoprecipitation for enhanced recovery of intact nuclear protein complexes. Antibody Binding Beads sold separately.
This kit improves Co-IP by using protein G-coated magnetic beads, speeding up and simplifying the procedure while greatly reducing background. Use with both nuclear & whole-cell extracts.

