THIS PRODUCT IS DISCONTINUED
Fast Activated Cell-based ELISA (FACE™) Kits provide a simple, sensitive method for detecting protein phosphorylation directly in the cell, without making extracts or performing electrophoresis and membrane blotting. These 96-well, high-throughput assays are available in both colorimetric and chemiluminescent formats for over 20 different targets (see list at right). For complete details, click the FACE™ Method tab below.
FACE HSP27 Kits provide 96 rxns each of 2 antibodies that enable you to monitor and compare the levels of both phosphorylated and total HSP27 (Heat Shock Protein 27). The phospho-HSP27 antibody recognizes HSP27 only when phosphorylated at Ser82. The total-HSP27 antibody recognizes HSP27 regardless of its phosphorylation state. Click the HSP27 Info tab below for data and more information.
| Name | Format | Cat No. | Price | |
|---|---|---|---|---|
| FACE™ HSP27 | 1 x 96 rxns | 48350 | Discontinued | |
| 5 x 96 rxns | 48850 | Discontinued | ||
| FACE™ HSP27 Chemi | 1 x 96 rxns | 48450 | Discontinued | |
| 5 x 96 rxns | 48950 | Discontinued | ||
| FACE™ HSP27 Manual |
| FACE™ Profile |
| Cell Biology Products Brochure |
| IsoCyte™ Application Note – Phospho-Protein Detection |
| MSDS: Sodium Azide |
| MSDS: Sulphuric Acid |
| MSDS: Thimersol |
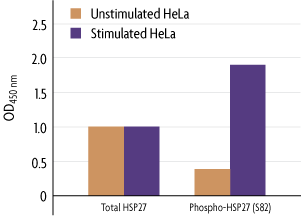
Figure 1: Measurement of phosphorylated HSP27 (S82) and total HSP27.
Human HeLa cells were cultured in 96-well plates and serum-starved (0.5% FBS) for 16 hours. Cells were then stimulated with 25 µg/ml of Anisomycin for 30 minutes and fixed. Total HSP27 and HSP27 phosphorylated on Ser82 were each assayed in duplicate using the phospho-HSP27 (S82) and total-HSP27 antibodies included in the FACE HSP27 Kit. Data were plotted after correction for cell number (performed through use of Crystal Violet).
Antibody Specificities
The phospho-HSP27 (Ser82) antibody was raised against a synthetic phospho-peptide corresponding to residues surrounding phosphorylated Ser 82 of human HSP27. The phospho-HSP27 (Ser82) antibody recognizes HSP27 only when phosphorylated at Ser82. The total-HSP27 antibody recognizes HSP27 protein regardless of its phosphorylation state.
HSP27 Overview
In response to stress, the expression level of HSP27 increases several fold to confer cellular resistance to the adverse environmental change. HSP27 is localized to the cytoplasm of unstressed cells but can redistribute to the nucleus in response to stress, where it may function to stabilize DNA and/or the nuclear membrane. HSP27 is phosphorylated at serines 15, 78 and 82 by MAPKAPK-2 as a result of the activation of the p38 MAPK pathway. Phosphorylation of HSP27 causes a change in its tertiary structure, which shifts from large homotypic multimers to dimers and monomers. Thus, phosphorylated HSPs have significantly decreased ability to act as molecular chaperones. Phosphorylation of HSP27 on Ser82 by MAPKAPK-2 or AKT leads to HSP dissociation from the AKT/MAPKAPK2/p38 complex, which may promote independent survival signals. The phosphorylation of HSP27 also enhances its association with IKKβ which results in decreased IKK activity.
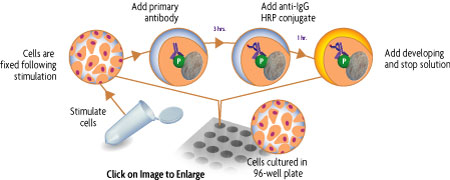
The FACE™ Method
In FACE, cells are cultured in 96-well plates and stimulated to induce the pathway of interest. Following stimulation, the cells are fixed rapidly, which preserves activation-specific protein modifications. Each well is then incubated with a primary antibody specific for the activated protein of interest. Subsequent incubation with secondary HRP-conjugated antibody and developing solution provides a colorimetric or chemiluminescent readout that is quantitative and reproducible (Figure 1). The number of cells in each well can be normalized easily with the provided Crystal Violet solution. FACE Kits also contain primary antibody specific for the native inactive protein, so you can monitor both native and activated protein levels in the same experiment. FACE eliminates cellular extractions, radioactive kinase assays, time-consuming Westerns and inefficient epitope interactions that occur on membranes. FACE is a highly sensitive high-throughput assay designed for detecting activated proteins within mammalian cells.
Figure 1: Flow chart of the FACE process.
Flow chart of the FACE in cell Western method that uses a cell based ELISA to measure the levels of the native and phospho forms of signaling proteins and kinases that are activated by phosphorylation.
Contents & Storage
Two (or ten) 96-well plates for culturing cells, 96 (or 5 x 96) rxns each of two primary antibodies (1 phospho-specific, 1 specific for native protein), HRP-conjugated secondary antibody, Quenching Solution, 1X Antibody Blocking Buffer, 1X Antibody Dilution Buffer, 10X PBS, 10% Triton X-100, 1% SDS Solution, Developing and Stop Solutions, and Crystal Violet Cell Quantification Solution. Storage conditions vary from room temperature to -20°C, see manual for details. All reagents are guaranteed stable for 6 months when stored properly.